|
11/28/2023 0 Comments Vaccine production us
Together with our partners, Moderna is committed to continuously developing best-in-class variant boosters so we can end this pandemic as fast as possible.”Īs shared at its Vaccines Day presentation on April 14, 2021, Moderna believes that this investment in increased supply is necessary due to an expected significant need for booster vaccinations in 2022 and beyond. 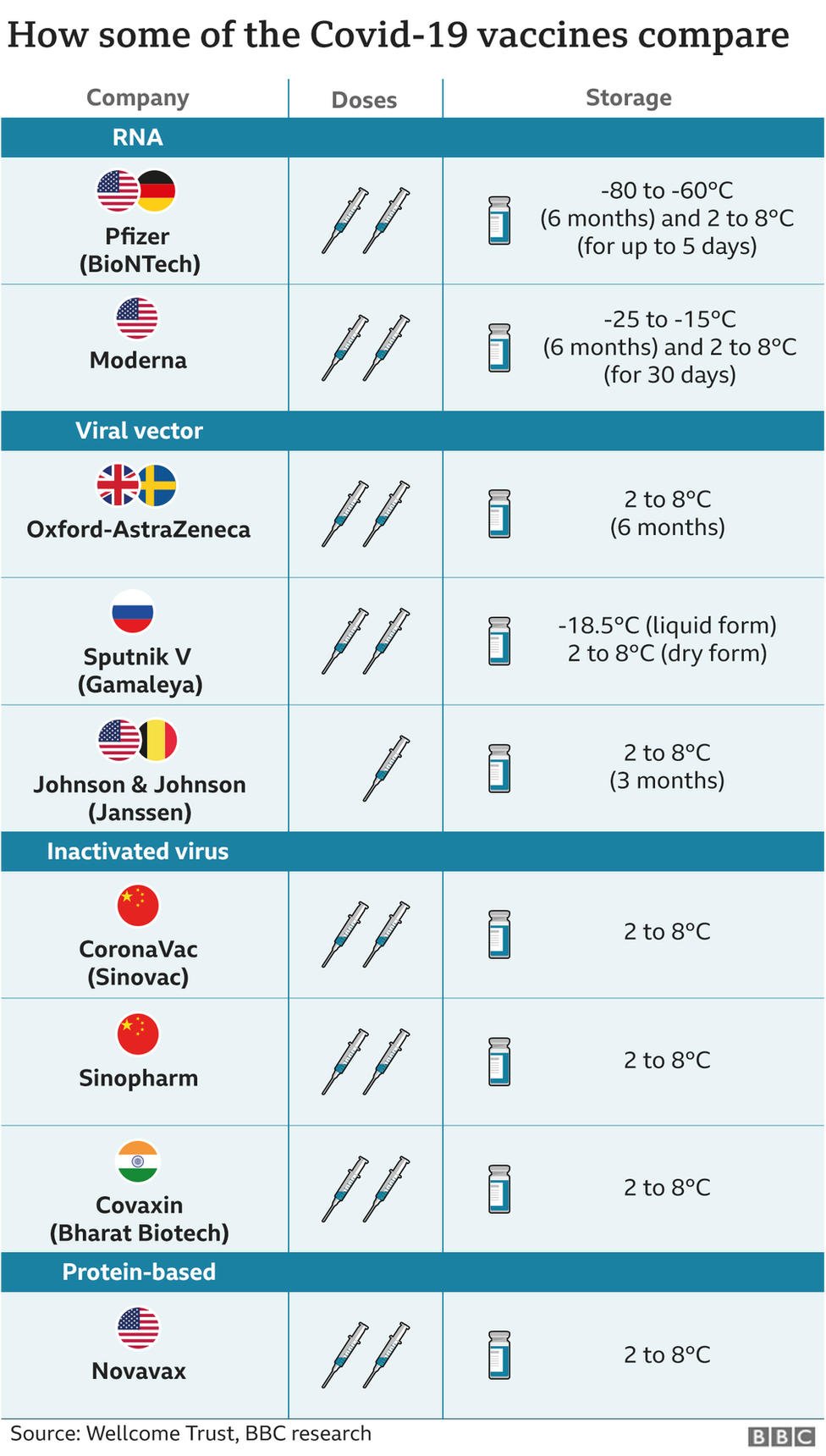
“We thank our manufacturing partners for their work and their commitment. will allow us to deliver up to 3 billion doses in 2022, depending on the mix of product between primary series of vaccination and variant boosters,” said Stéphane Bancel, Chief Executive Officer of Moderna. Today we have announced that our investments in Europe, including Spain, France and Belgium, Switzerland, and the U.S. We are hearing from governments that there is no technology that provides the high efficacy of mRNA vaccines and the speed necessary to adapt to variants, while allowing reliable scalability of manufacturing. “As we follow the rapid spread of SARS-CoV-2 variants of concern, we believe that there will continue to be significant need for our mRNA COVID-19 vaccine and our variant booster candidates into 20. 
In addition, Moderna is in advanced negotiations for other agreements. The increases announced today are in addition to the recently announced increases in formulation, fill and finish in the U.S. Today, the Company also raised its 2021 manufacturing supply forecast to between 800 million to 1 billion doses. The Company will begin making investments at its owned and partnered manufacturing facilities in 2021, with increased production from these investments expected to ramp up in late 2021 and early 2022. When completed, the investments will also result in an increase in safety stock of raw materials and finished product used to deliver committed volumes. These investments allow for a doubling of drug substance manufacturing at Lonza’s (SIX: LONN) Switzerland-based facility, a more than doubling of formulation, fill and finish and drug substance manufacturing at Rovi’s (BME: ROVI) Spain-based facility, as well as a 50% increase of drug substance at Moderna’s facilities in the U.S. The Company will use its cash balance to fund these investments. (Nasdaq: MRNA), a biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines, today announced it is making new funding commitments to increase supply at its owned and partnered manufacturing facilities, which it expects will increase global 2022 capacity to up to 3 billion doses of its COVID-19 vaccine, depending upon the mix between the authorized Moderna COVID-19 Vaccine at the 100 μg dose level and potentially lower doses of the Company’s variant booster candidates and pediatric vaccines, if authorized. Supply also expected to benefit from shift to lower-dose product mix, including potential variant booster vaccines and pediatric primary vaccine dosesĬompany increases its 2021 supply forecast to between 800 million and 1 billion dosesĬompany also announced data supporting 3-month refrigerated (2-8° C) stable formulationĬAMBRIDGE, Mass.-(BUSINESS WIRE)-Apr. Investments to increase supply globally, including doubling supply from Moderna’s ex- U.S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
